IP3 receptor-mediated Ca2+ release from acidocalcisomes regulates mitochondrial bioenergetics and prevents autophagy in Trypanosoma cruzi
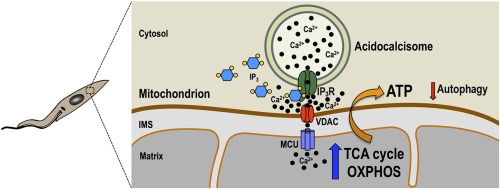
 In contrast to animal cells, the inositol 1,4,5-trisphosphate receptor of Trypanosoma cruzi (TcIP3R) localizes to acidocalcisomes instead of the endoplasmic reticulum. Here, we present evidence that TcIP3R is a Ca2+ release channel gated by IP3 when expressed in DT40 cells knockout for all vertebrate IP3 receptors, and is required for Ca2+ uptake by T. cruzi mitochondria, regulating pyruvate dehydrogenase dephosphorylation and mitochondrial O2 consumption, and preventing autophagy. Localization studies revealed its co-localization with an acidocalcisome marker in all life cycle stages of the parasite. Ablation of TcIP3R by CRISPR/Cas9 genome editing caused: a) a reduction in O2 consumption rate and citrate synthase activity; b) decreased mitochondrial Ca2+ transport without affecting the membrane potential; c) increased ammonia production and AMP/ATP ratio; d) stimulation of autophagosome formation, and e) marked defects in growth of culture forms (epimastigotes) and invasion of host cells by infective stages (trypomastigotes). Moreover, TcIP3R overexpressing parasites showed decreased metacyclogenesis, trypomastigote host cell invasion and intracellular amastigote replication. In conclusion, the results suggest a modulatory activity of TcIP3R-mediated acidocalcisome Ca2+ release on cell bioenergetics in T. cruzi.
In contrast to animal cells, the inositol 1,4,5-trisphosphate receptor of Trypanosoma cruzi (TcIP3R) localizes to acidocalcisomes instead of the endoplasmic reticulum. Here, we present evidence that TcIP3R is a Ca2+ release channel gated by IP3 when expressed in DT40 cells knockout for all vertebrate IP3 receptors, and is required for Ca2+ uptake by T. cruzi mitochondria, regulating pyruvate dehydrogenase dephosphorylation and mitochondrial O2 consumption, and preventing autophagy. Localization studies revealed its co-localization with an acidocalcisome marker in all life cycle stages of the parasite. Ablation of TcIP3R by CRISPR/Cas9 genome editing caused: a) a reduction in O2 consumption rate and citrate synthase activity; b) decreased mitochondrial Ca2+ transport without affecting the membrane potential; c) increased ammonia production and AMP/ATP ratio; d) stimulation of autophagosome formation, and e) marked defects in growth of culture forms (epimastigotes) and invasion of host cells by infective stages (trypomastigotes). Moreover, TcIP3R overexpressing parasites showed decreased metacyclogenesis, trypomastigote host cell invasion and intracellular amastigote replication. In conclusion, the results suggest a modulatory activity of TcIP3R-mediated acidocalcisome Ca2+ release on cell bioenergetics in T. cruzi.
Miguel A. Chiurillo, Noelia Lander, Anibal E. Vercesi, Roberto Docampo, 2020. Cell Calcium; 92:102284, https://doi.org/10.1016/j.ceca.2020.102284.
