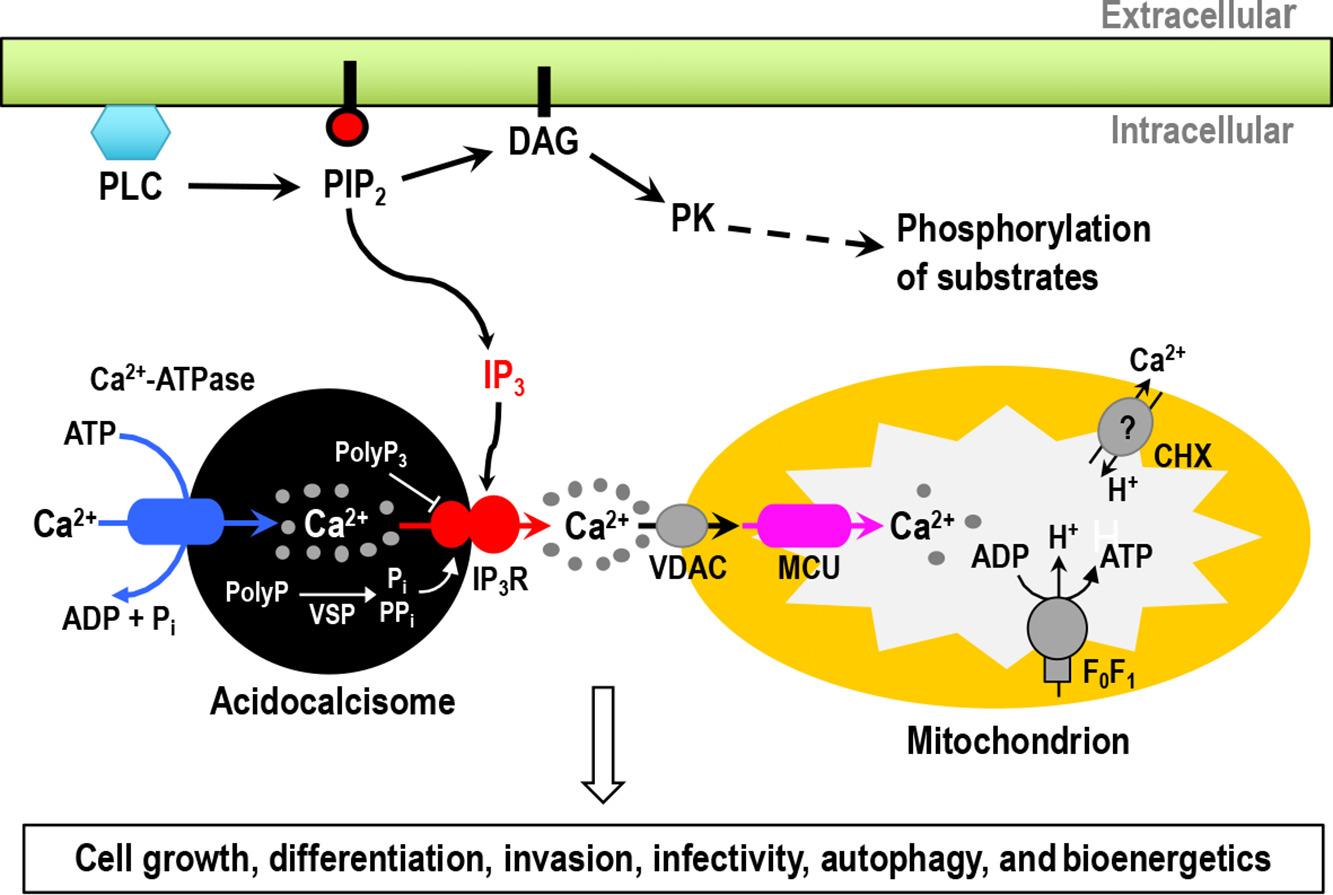
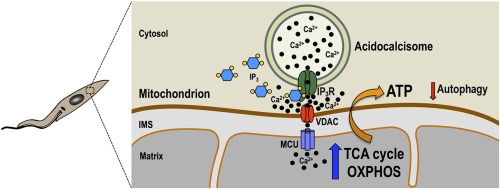
TbVps41 regulates trafficking of endocytic but not biosynthetic cargo to lysosomes of bloodstream forms of Trypanosoma brucei
The bloodstream stage of Trypanosoma brucei, the causative agent of African trypanosomiasis, is characterized by its high rate of endocytosis, which is involved in remodeling of its surface coat. Here we present evidence that RNAi-mediated expression down-regulation of vacuolar protein sorting 41 (Vps41), a component of the homotypic fusion and vacuole protein sorting (HOPS) complex, …