Validation of a multiplex microsphere immunoassay for detection of antibodies to Trypanosoma cruzi in dogs
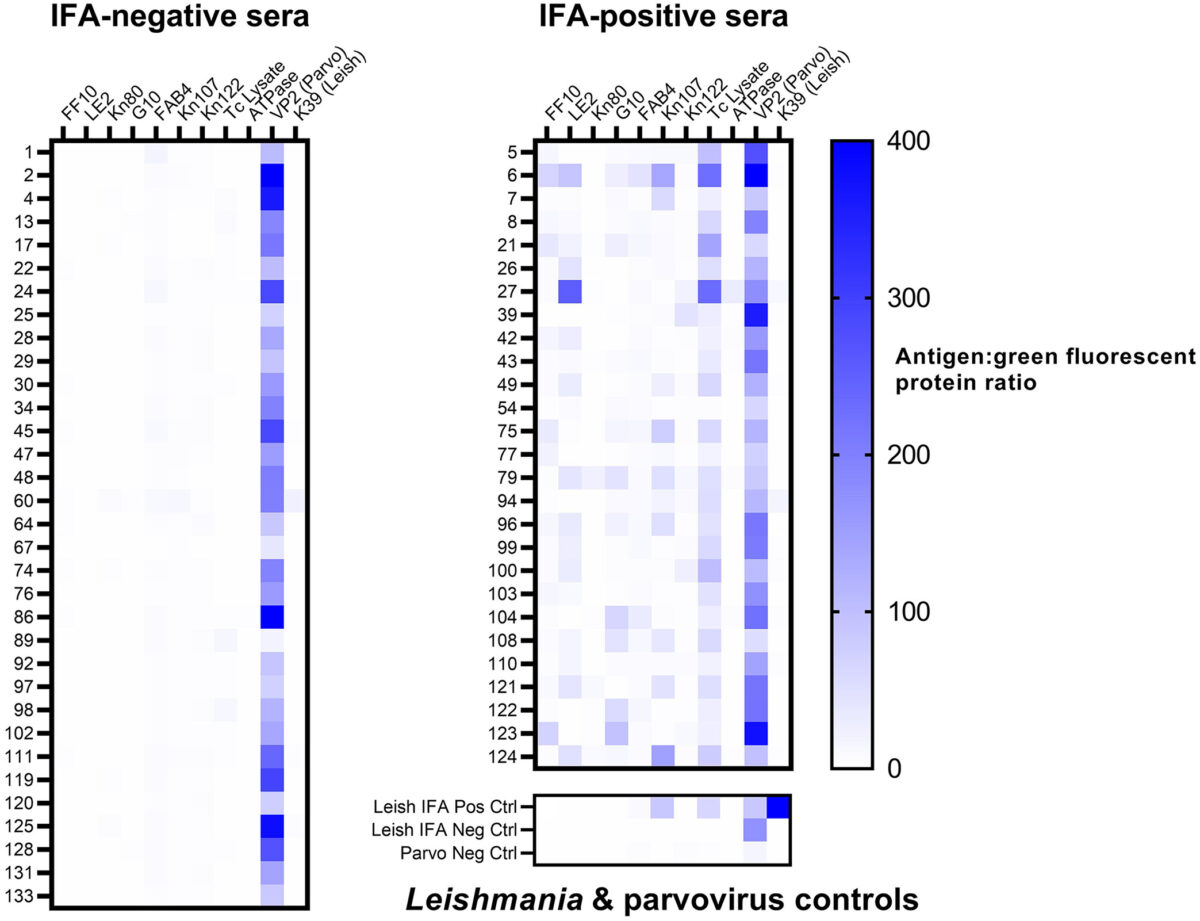
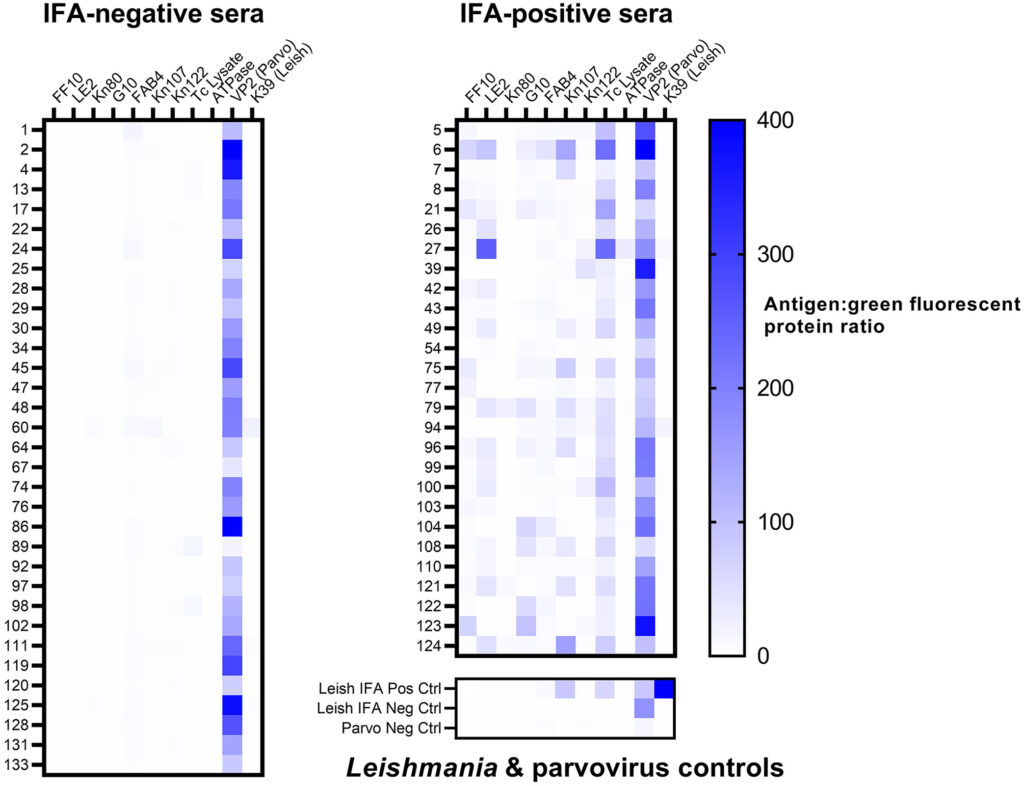
The vector-borne protozoan parasite Trypanosoma cruzi causes Chagas disease in humans, dogs, and many other mammalian hosts. Canine Chagas disease is increasingly diagnosed in dogs of the southern United States where triatomine insect vectors occur, and there are limited veterinary testing options; only the indirect fluorescent antibody (IFA) test is offered at a single accredited diagnostic laboratory. We evaluated a multiplex microsphere immunoassay (MIA) for the detection of antibodies against T. cruzi in dogs and compared it with existing serologic methods to establish cutoff values and relative sensitivity and specificity. We tested 135 canine sera that had been characterized using the IFA and off-label use of 2 commercial rapid assays with our multiplex MIA against 12 antigens: 9 T. cruzi antigens, a negative control recombinant protein (green fluorescent protein, GFP), a Leishmania antigen, and a canine parvovirus antigen (used as an antibody control given near-ubiquitous parvoviral vaccination). The median fluorescence intensity (MFI) ratio between each T. cruzi antigen and GFP was calculated for every sample. Samples with an antigen:GFP MFI ratio > 4 SDs above the mean of 25 known-negative sera were considered positive to that antigen. Samples testing positive to ≥ 2 antigens were considered positive for T. cruzi antibodies. Compared to the IFA, our multiplex MIA had a relative sensitivity of 100% and specificity of 97.0%. Given its precision, high-throughput format, potential for automation, and lack of subjective interpretation, our multiplex MIA should be considered a valid and improved assay for T. cruzi antibodies in dogs.
Carlos A Rodriguez, Rachel E Busselman, Huifeng Shen, Ashley B Saunders, Rick Tarleton, Sarah A Hamer. J Vet Diagn Invest. 2023 Sep 5;10406387231198525. doi: 10.1177/10406387231198525.
