ANTI-MALARIAL ACTIVITY OF AMENTOFLAVONE ISOLATED FROM LEAF OF CALOPHYLLUM TOMENTOSUM WEIGHT
Calophyllum tomentosum belonging to Clusiaceae family is an Indian medicinal plant used as folklore medicine to cure various kinds of diseases reported in Ayurveda, and the leaves of the plant are also used as an active ingredient for the preparation of a botanical medicine known as ‘Punnaga’, ‘Surapunnaga’ and ‘Tamoil’ among other common names. Chemical …
Researchers discover malaria gene needed to make pair of invasion organelles
Optimization of diastereomeric dihydropyridines as antimalarials
The increase in research funding for the development of antimalarials since 2000 has led to a surge of new chemotypes with potent antimalarial activity. High-throughput screens have delivered several thousand new active compounds in several hundred series, including the 4,7-diphenyl-1,4,5,6,7,8-hexahydroquinolines, hereafter termed dihydropyridines (DHPs). We optimized the DHPs for antimalarial activity. Structure-activity relationship studies focusing …
The influence of oviposition status on measures of transmission potential in malaria-infected mosquitoes depends on sugar availability
Background: Like other oviparous organisms, the gonotrophic cycle of mosquitoes is not complete until they have selected a suitable habitat to oviposit. In addition to the evolutionary constraints associated with selective oviposition behavior, the physiological demands relative to an organism’s oviposition status also influence their nutrient requirement from the environment. Yet, studies that measure transmission potential …
Trainee Spotlight: Grace Vick
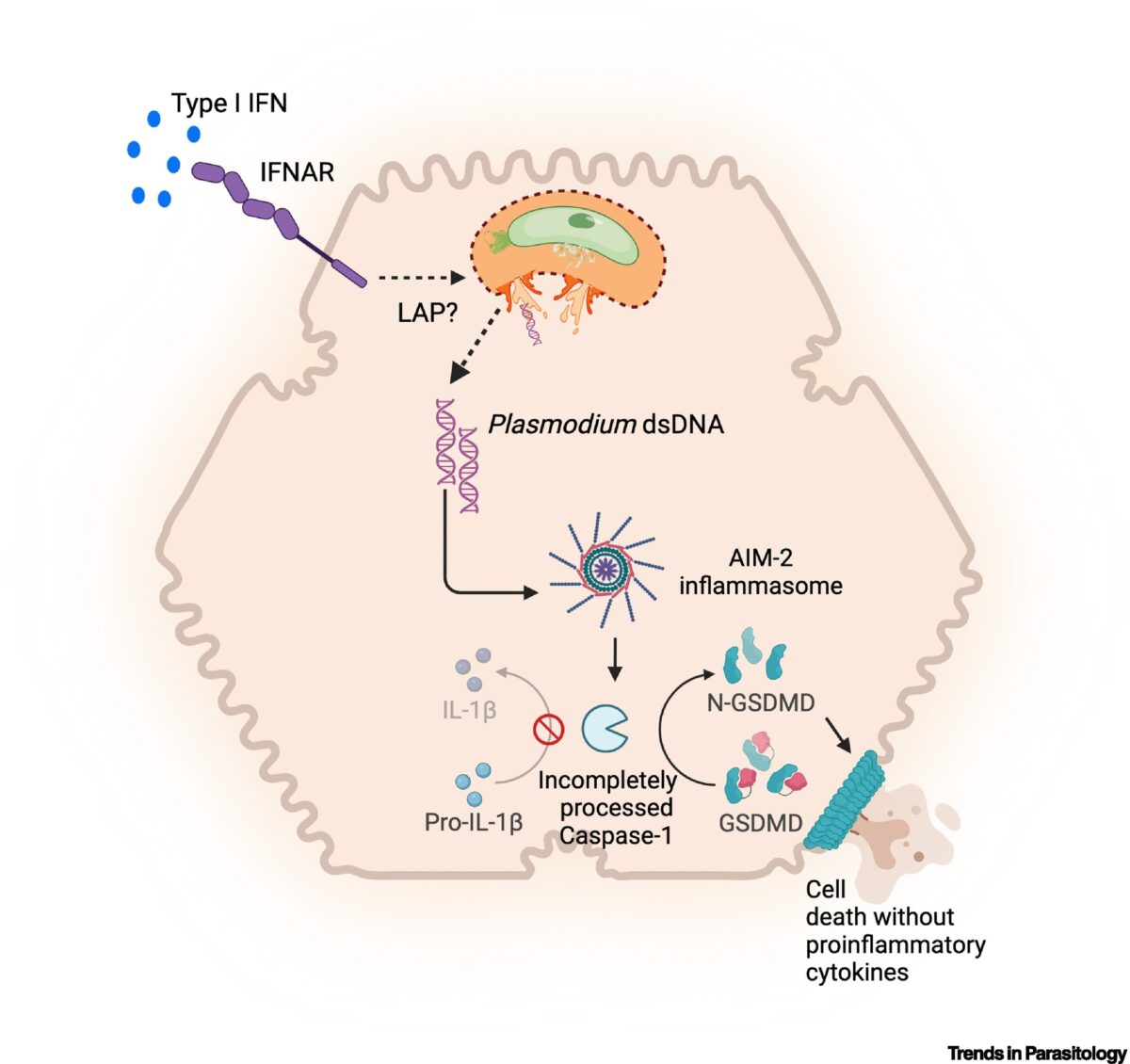
Hepatocytes and the art of killing Plasmodium softly
The Plasmodium parasites that cause malaria undergo asymptomatic development in the parenchymal cells of the liver, the hepatocytes, prior to infecting erythrocytes and causing clinical disease. Traditionally, hepatocytes have been perceived as passive bystanders that allow hepatotropic pathogens such as Plasmodium to develop relatively unchallenged. However, now there is emerging evidence suggesting that hepatocytes …
Aptamer-Based Imaging of Polyisoprenoids in the Malaria Parasite
Dolichols are isoprenoid end-products of the mevalonate and 2C-methyl-D-erythritol-4-phosphate pathways. The synthesis of dolichols is initiated with the addition of several molecules of isopentenyl diphosphate to farnesyl diphosphate. This reaction is catalyzed by a cis-prenyltransferase and leads to the formation of polyprenyl diphosphate. Subsequent steps involve the dephosphorylation and reduction of the α-isoprene unit …