Development and Introduction of the Filariasis Test Strip: A New Diagnostic Test for the Global Program to Eliminate Lymphatic Filariasis
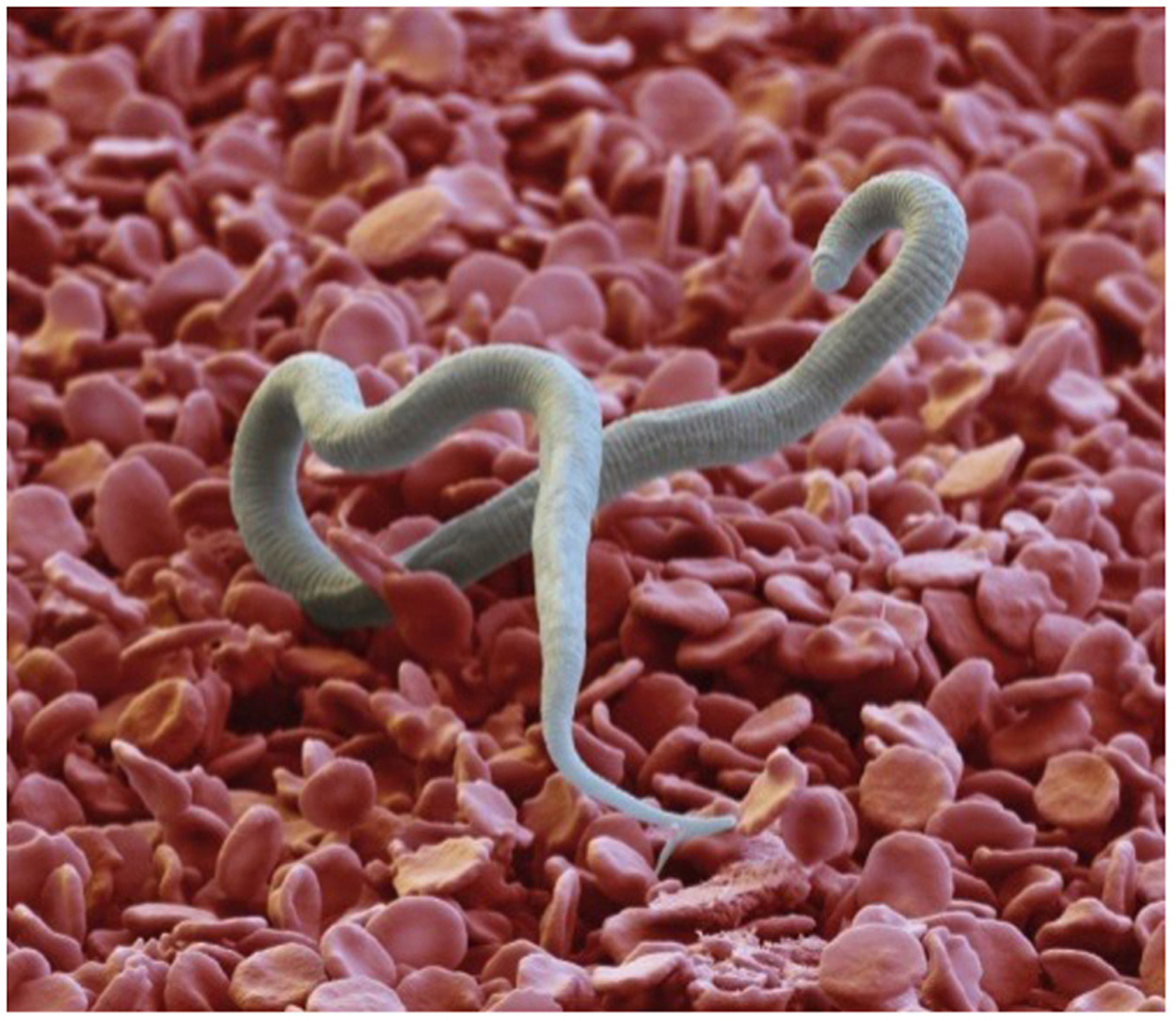
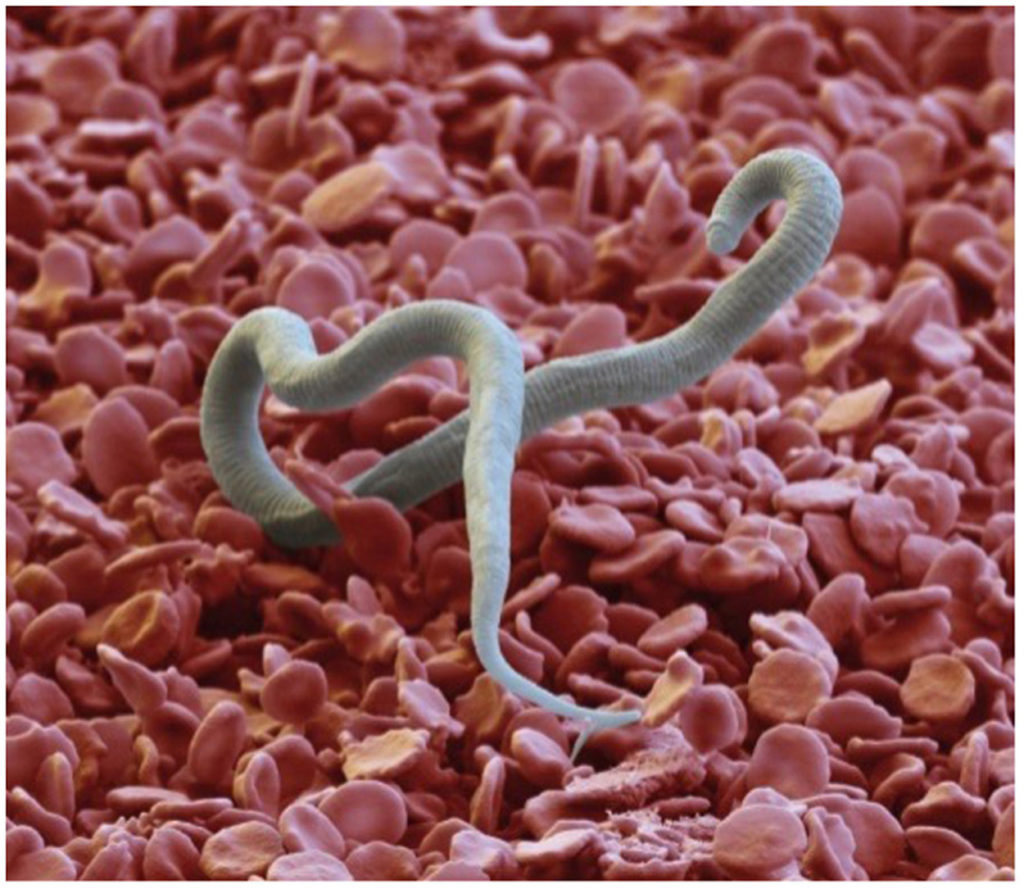
A key component to achieving the global goal of elimination of lymphatic filariasis (LF) is the availability of appropriate tools for disease mapping, monitoring, and surveillance. However, the development of these tools for a neglected disease such as LF can be a challenge. The lack of a commercial market and low familiarity with these diseases leave little incentive for diagnostic manufacturers to invest in this space. The Filarial Test Strip (FTS) development story provides a case study on how a multi-stakeholder, public-private partnership model facilitated the development, evaluation, and introduction of a new monitoring and surveillance tool for LF. This paper will reflect on the experience with the FTS and document the process from development of the target product profile to adoption and scale-up in country programs. Lessons learned from both the successes and challenges experienced during this process may help inform future efforts to develop and introduce new diagnostic or surveillance tools for neglected diseases.
Anastasia Pantelias, Jonathan D King, Patrick Lammie, Gary J Weil. Am J Trop Med Hyg. 2022 Mar 15;tpmd210990. doi: 10.4269/ajtmh.21-0990.