Probing the B- & C-rings of the antimalarial tetrahydro-β-carboline MMV008138 for steric and conformational constraints
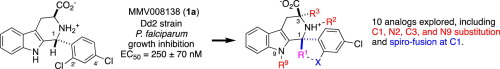
The antimalarial candidate MMV008138 (1a) is of particular interest because its target enzyme (IspD) is absent in human. To achieve higher potency, and to probe for steric demand, a series of analogs of 1a were prepared that featured methyl-substitution of the B- and C-rings, as well as ring-chain transformations. X-ray crystallography, NMR spectroscopy and calculation were used to study the effects of these modifications on the conformation of the C-ring and orientation of the D-ring. Unfortunately, all the B- and C-ring analogs explored lost in vitro antimalarial activity. The possible role of steric effects and conformational changes on target engagement are discussed.
Sha Ding, Maryam Ghavami, Joshua H.Butler, Emilio F. Merino, Carla Slebodnick, Maria B. Cassera, Paul R. Carlier. Bioorg Med Chem Lett. 2020 Sep 5;127520. doi: 10.1016/j.bmcl.2020.127520
