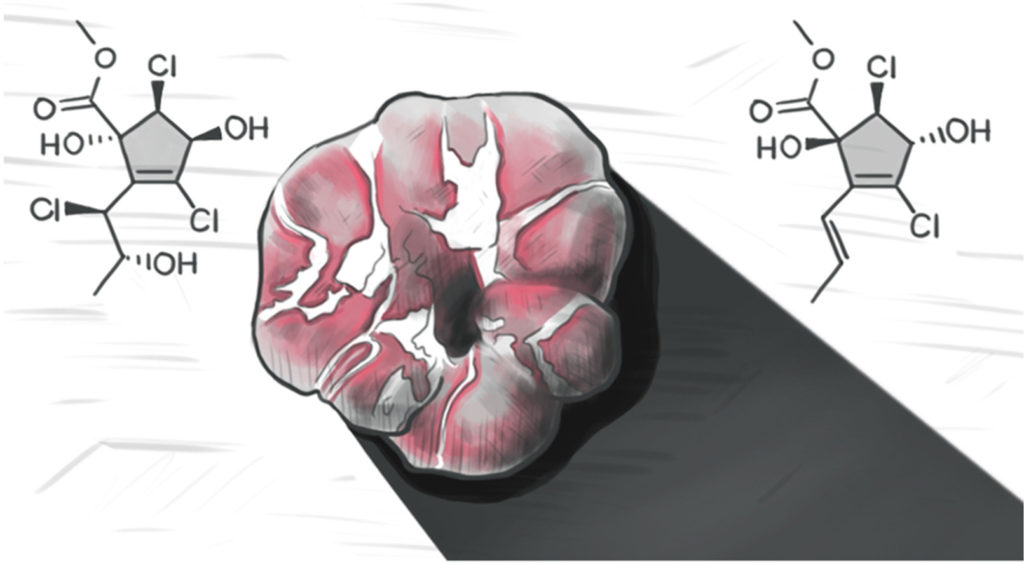
Polychlorinated cyclopentenes from a marine derived Periconia sp. (strain G1144)
Studies on an organic extract of a marine fungus, Periconia sp. (strain G1144), led to the isolation of three halogenated cyclopentenes along with the known and recently reported rhytidhyester D; a series of spectrometric and spectroscopic techniques were used to elucidate these structures. Interestingly, two of these compounds represent tri-halogenated cyclopentene derivatives, which have been observed only rarely from Nature. The relative and absolute configurations of the compounds were established via mass spectrometry (MS), nuclear magnetic resonance (NMR) spectroscopy, Mosher’s esters method, optical rotation and GIAO NMR calculations, including correlation coefficient calculations and the use of both DP4+ and dJ DP4 analyses. Several of the isolated compounds were tested for activity in anti-parasitic, antimicrobial, quorum sensing inhibition, and cytotoxicity assays and were shown to be inactive.
Kristóf B Cank, Robert A Shepherd, Sonja L Knowles, Manuel Rangel-Grimaldo, Huzefa A Raja, Zoie L Bunch, Nadja B Cech, Christopher A Rice, Dennis E Kyle, Joseph O Falkinham 3rd, Joanna E Burdette, Nicholas H Oberlies. Phytochemistry . 2022 Apr 11;113200. doi: 10.1016/j.phytochem.2022.113200