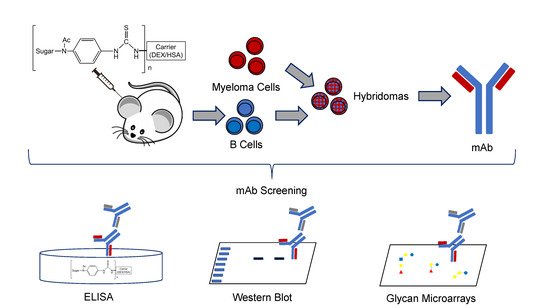
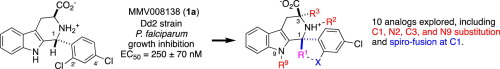
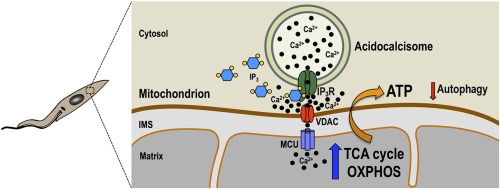
Dennis Kyle: Finding Solutions for Deadly Diseases
Dennis Kyle leads the UGA Center for Tropical and Emerging Diseases, and his endowment enables him to run a 16-person lab of student researchers, postdocs, and research scientists fighting a host of parasitic diseases around the world. (Photo by Andrew Davis Tucker/UGA) GRA Endowment helps researchers save lives through drug discovery The Amoeba Summit in …