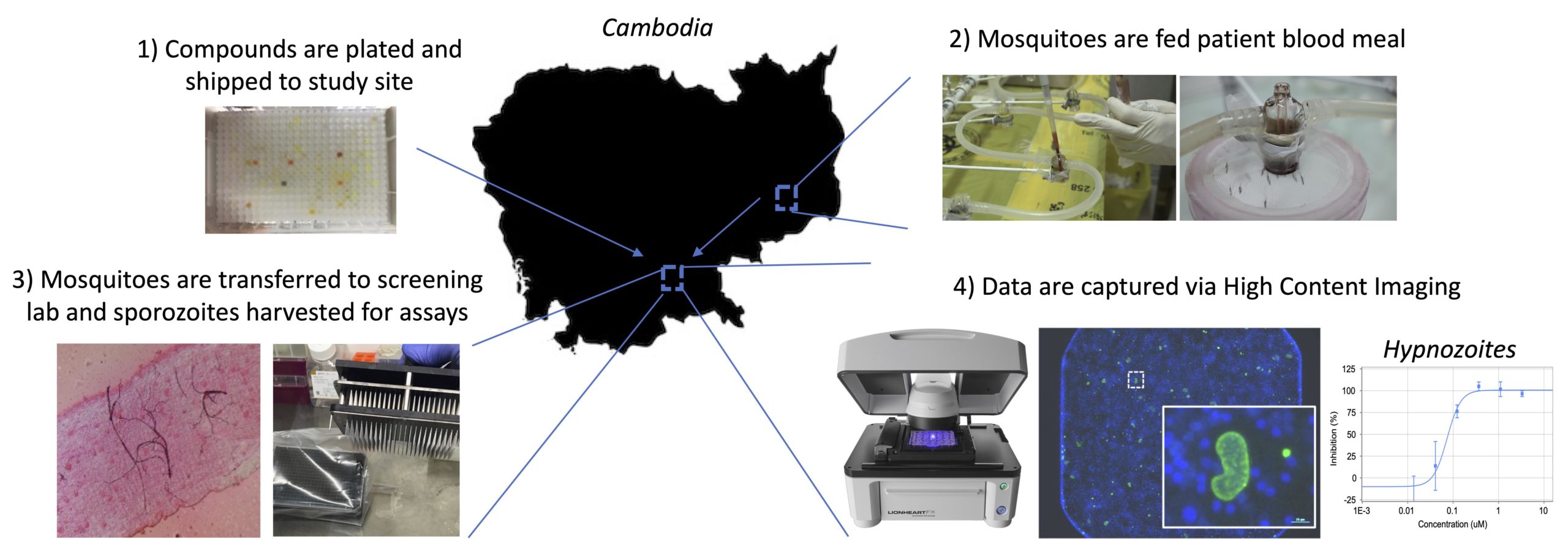
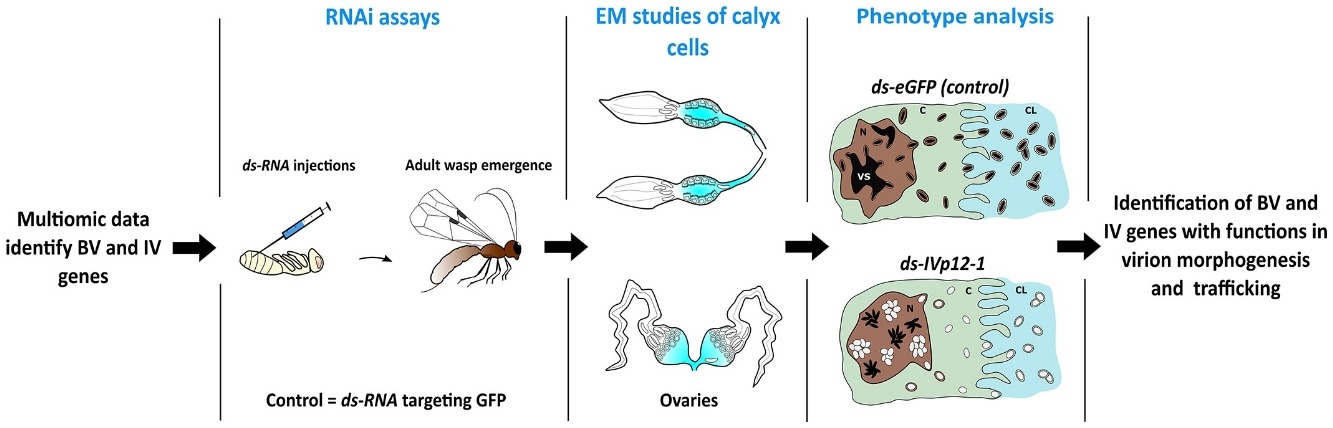
Antimalarial Natural Products
Natural products have made a crucial and unique contribution to human health, and this is especially true in the case of malaria, where the natural products quinine and artemisinin and their derivatives and analogues, have saved millions of lives. The need for new drugs to treat malaria is still urgent, since the most dangerous malaria …