An Endoplasmic Reticulum CREC Family Protein Regulates the Egress Proteolytic Cascade in Malaria Parasites
The endoplasmic reticulum (ER) is thought to play an essential role during egress of malaria parasites because the ER is assumed to be required for biogenesis and secretion of egress-related organelles. However, no proteins localized to the parasite ER have been shown to play a role in egress of malaria parasites. In this study, we generated conditional mutants of the Plasmodium falciparum endoplasmic reticulum-resident calcium-binding protein (PfERC), a member of the CREC family. Knockdown of the PfERC gene showed that this gene is essential for asexual growth of P. falciparum Analysis of the intraerythrocytic life cycle revealed that PfERC is essential for parasite egress but is not required for protein trafficking or calcium storage. We found that PfERC knockdown prevents the rupture of the parasitophorous vacuole membrane. This is because PfERC knockdown inhibited the proteolytic maturation of the subtilisin-like serine protease SUB1. Using double mutant parasites, we showed that PfERC is required for the proteolytic maturation of the essential aspartic protease plasmepsin X, which is required for SUB1 cleavage. Further, we showed that processing of substrates downstream of the proteolytic cascade is inhibited by PfERC knockdown. Thus, these data establish that the ER-resident CREC family protein PfERC is a key early regulator of the egress proteolytic cascade of malaria parasites.
IMPORTANCE The divergent eukaryotic parasites that cause malaria grow and divide within a vacuole inside a host cell, which they have to break open once they finish cell division. The egress of daughter parasites requires the activation of a proteolytic cascade, and a subtilisin-like protease initiates a proteolytic cascade to break down the membranes blocking egress. It is assumed that the parasite endoplasmic reticulum plays a role in this process, but the proteins in this organelle required for egress remain unknown. We have identified an early ER-resident regulator essential for the maturation of the recently discovered aspartic protease in the egress proteolytic cascade, plasmepsin X, which is required for maturation of the subtilisin-like protease. Conditional loss of PfERC results in the formation of immature and inactive egress proteases that are unable to breakdown the vacuolar membrane barring release of daughter parasites.
Manuel A. Fierro, Beejan Asady, Carrie F. Brooks, David W. Cobb, Alejandra Villegas, Silvia N. J. Moreno, Vasant Muralidharan. mBio. 2020 Feb 25;11(1). pii: e03078-19. doi: 10.1128/mBio.03078-19.

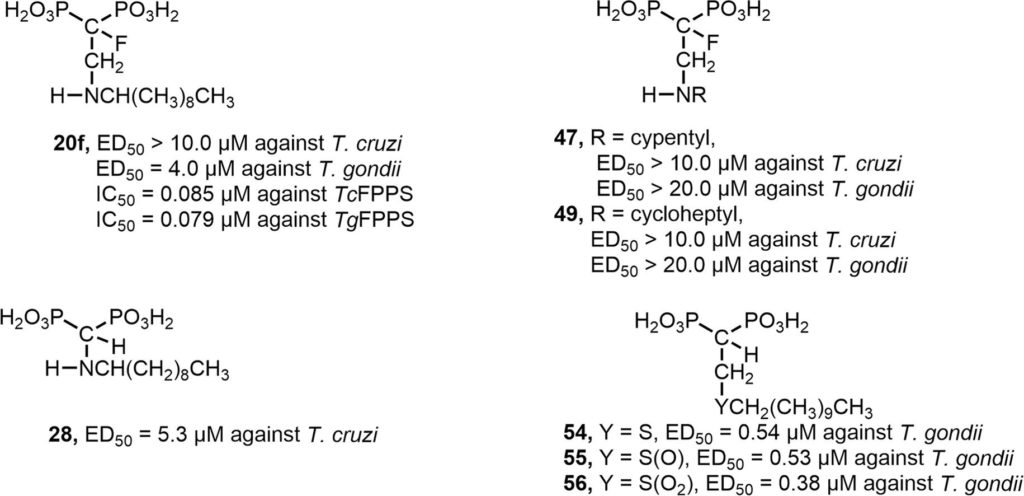
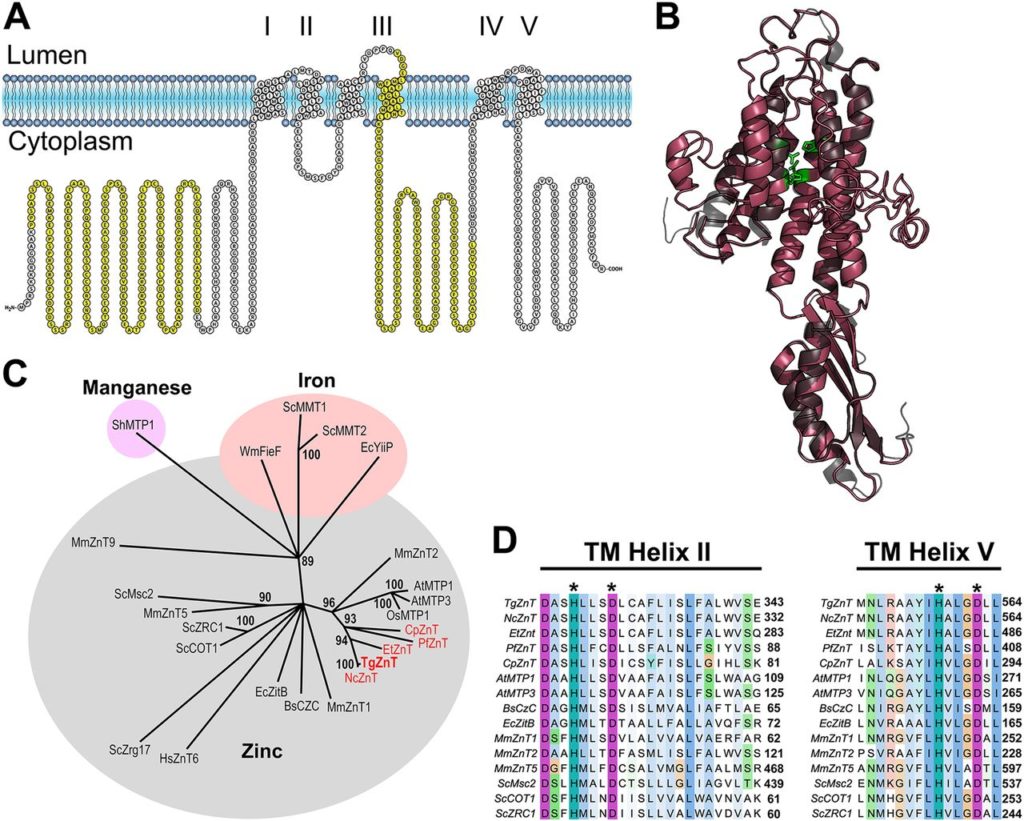 Zinc (Zn2+) is the most abundant biological metal ion aside from iron and is an essential element in numerous biological systems, acting as a cofactor for a large number of enzymes and regulatory proteins. Zn2+ must be tightly regulated, as both the deficiency and overabundance of intracellular free Zn2+ are harmful to cells. Zn2+ transporters (ZnTs) play important functions in cells by reducing intracellular Zn2+ levels by transporting the ion out of the cytoplasm. We characterized a
Zinc (Zn2+) is the most abundant biological metal ion aside from iron and is an essential element in numerous biological systems, acting as a cofactor for a large number of enzymes and regulatory proteins. Zn2+ must be tightly regulated, as both the deficiency and overabundance of intracellular free Zn2+ are harmful to cells. Zn2+ transporters (ZnTs) play important functions in cells by reducing intracellular Zn2+ levels by transporting the ion out of the cytoplasm. We characterized a 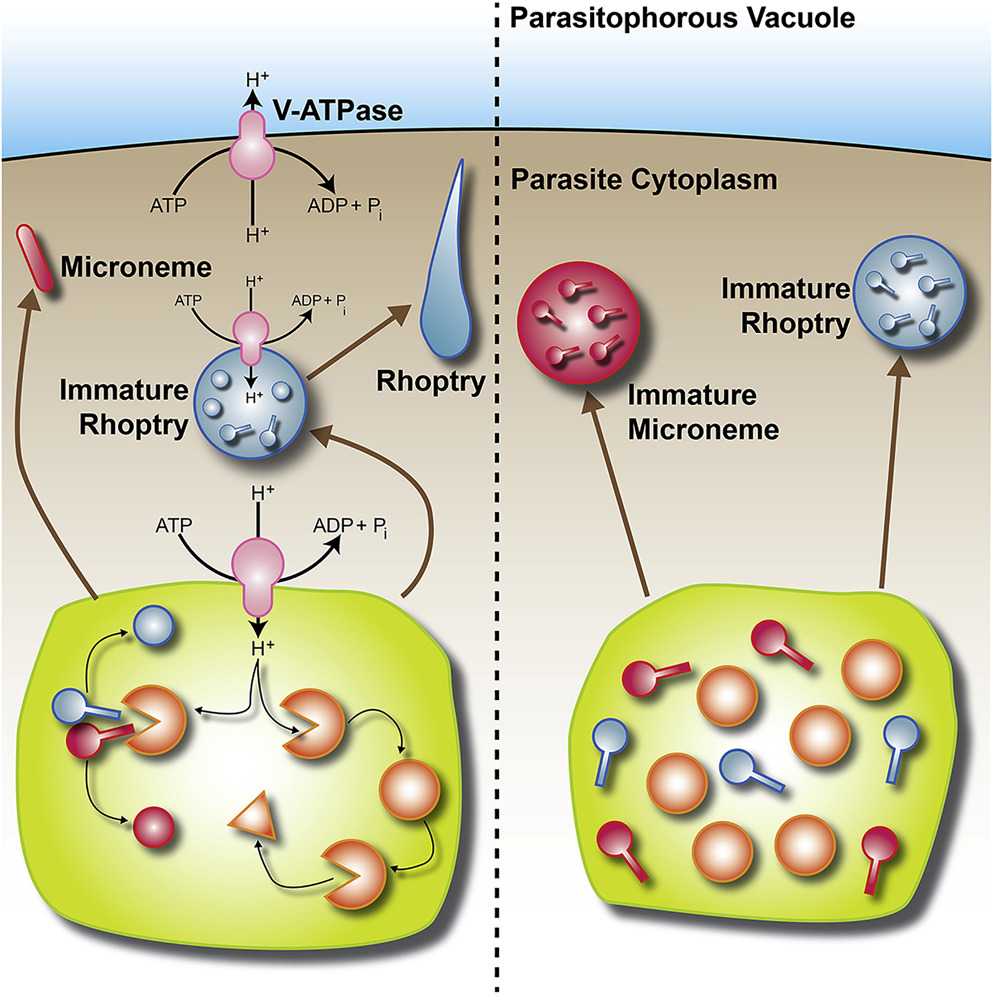 Vacuolar-proton ATPases (V-ATPases) are conserved complexes that couple the hydrolysis of ATP to the pumping of protons across membranes. V-ATPases are known to play diverse roles in cellular physiology. We studied the Toxoplasma gondiiV-ATPase complex and discovered a dual role of the pump in protecting parasites against ionic stress and in the maturation of secretory proteins in endosomal-like compartments. Toxoplasma V-ATPase subunits localize to the plasma membrane and to acidic vesicles, and characterization of conditional mutants of the a1 subunit highlighted the functionality of the complex at both locations. Microneme and rhoptry proteins are required for invasion and modulation of host cells, and they traffic via endosome-like compartments in which proteolytic maturation occurs. We show that the V-ATPase supports the maturation of rhoptry and microneme proteins, and their maturases, during their traffic to their corresponding organelles. This work underscores a role for V-ATPases in regulating virulence pathways.
Vacuolar-proton ATPases (V-ATPases) are conserved complexes that couple the hydrolysis of ATP to the pumping of protons across membranes. V-ATPases are known to play diverse roles in cellular physiology. We studied the Toxoplasma gondiiV-ATPase complex and discovered a dual role of the pump in protecting parasites against ionic stress and in the maturation of secretory proteins in endosomal-like compartments. Toxoplasma V-ATPase subunits localize to the plasma membrane and to acidic vesicles, and characterization of conditional mutants of the a1 subunit highlighted the functionality of the complex at both locations. Microneme and rhoptry proteins are required for invasion and modulation of host cells, and they traffic via endosome-like compartments in which proteolytic maturation occurs. We show that the V-ATPase supports the maturation of rhoptry and microneme proteins, and their maturases, during their traffic to their corresponding organelles. This work underscores a role for V-ATPases in regulating virulence pathways.

