Oxygen-dependent regulation of E3(SCF)ubiquitin ligases and a Skp1-associated JmjD6 homolog in development of the social amoeba Dictyostelium
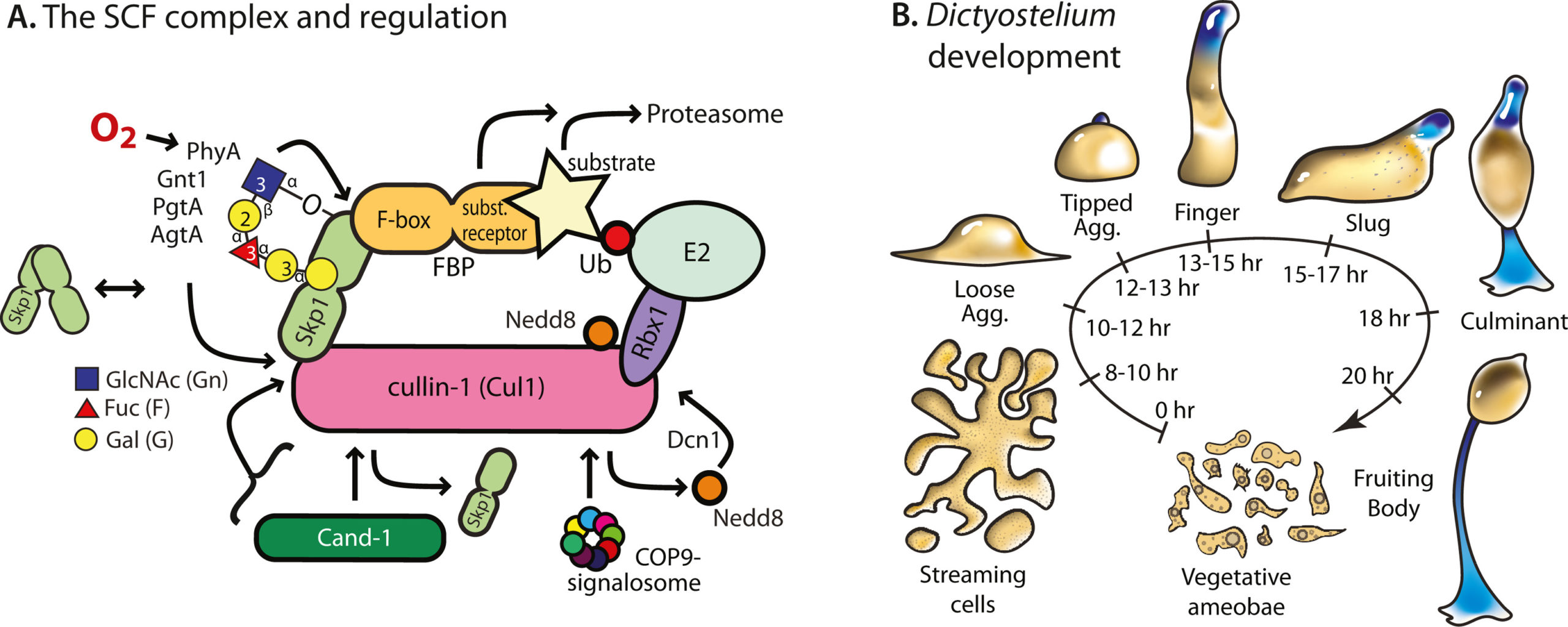
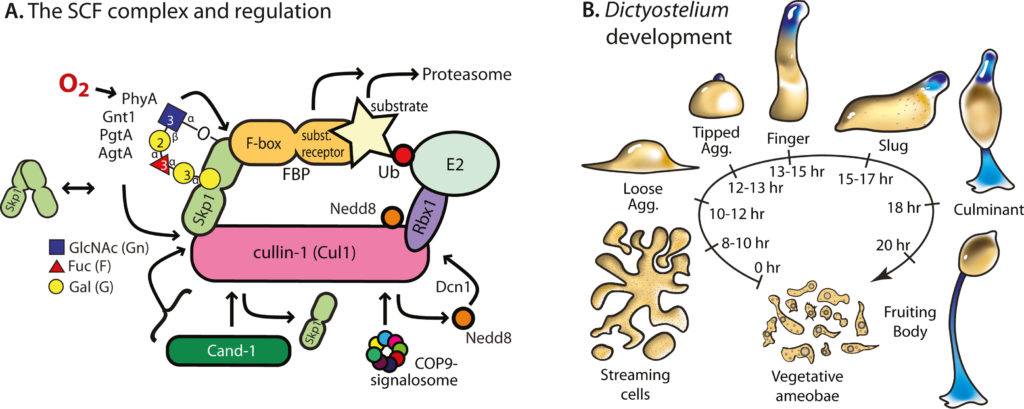 E3-SCF (Skp1/cullin-1/F-box protein) polyubiquitin ligases activate the proteasomal degradation of over a thousand proteins, but the evolutionary diversification of the F-box protein (FBP) family of substrate receptor subunits has challenged their elucidation in protists. Here we expand the FBP candidate list in the social amoeba Dictyostelium and show that the Skp1 interactome is highly remodeled as cells transition from solitary growth to multicellular development. Importantly, a subset of candidate FBPs was less represented when the posttranslational hydroxylation and glycosylation of Skp1 was abrogated by deletion of the O2 -sensing Skp1 prolyl hydroxylase PhyA. A role for this Skp1 modification for SCF activity was indicated by partial rescue of development, which normally depends on high O2 and PhyA, of phyA -knockout cells by proteasomal inhibitors. Further examination of two FBPs, FbxwD and the Jumonji C protein JcdI, suggested that Skp1 was substituted by other factors in phyA-knockout cells. Although a double-knockout of jcdI and its paralog jcdH did not affect development, overexpression of JcdI increased its sensitivity to O2. JcdI, a non-heme dioxygenase shown to have physiological O2-dependence, is conserved across protists with its F-box and other domains, and related to the human oncogene JmjD6. Sensitization of JcdI-overexpression cells to O2 depended on its dioxygenase activity and other domains, but not its F-box, which may however be the mediator of its reduced levels in wild-type relative to Skp1 modification mutant cells. The findings suggest that activation of JcdI by O2 is tempered by homeostatic down-regulation via PhyA and association with Skp1.
E3-SCF (Skp1/cullin-1/F-box protein) polyubiquitin ligases activate the proteasomal degradation of over a thousand proteins, but the evolutionary diversification of the F-box protein (FBP) family of substrate receptor subunits has challenged their elucidation in protists. Here we expand the FBP candidate list in the social amoeba Dictyostelium and show that the Skp1 interactome is highly remodeled as cells transition from solitary growth to multicellular development. Importantly, a subset of candidate FBPs was less represented when the posttranslational hydroxylation and glycosylation of Skp1 was abrogated by deletion of the O2 -sensing Skp1 prolyl hydroxylase PhyA. A role for this Skp1 modification for SCF activity was indicated by partial rescue of development, which normally depends on high O2 and PhyA, of phyA -knockout cells by proteasomal inhibitors. Further examination of two FBPs, FbxwD and the Jumonji C protein JcdI, suggested that Skp1 was substituted by other factors in phyA-knockout cells. Although a double-knockout of jcdI and its paralog jcdH did not affect development, overexpression of JcdI increased its sensitivity to O2. JcdI, a non-heme dioxygenase shown to have physiological O2-dependence, is conserved across protists with its F-box and other domains, and related to the human oncogene JmjD6. Sensitization of JcdI-overexpression cells to O2 depended on its dioxygenase activity and other domains, but not its F-box, which may however be the mediator of its reduced levels in wild-type relative to Skp1 modification mutant cells. The findings suggest that activation of JcdI by O2 is tempered by homeostatic down-regulation via PhyA and association with Skp1.
Andrew W Boland, Elisabet Gas-Pascual, Braxton L Nottingham, Hanke van der Wel, M Osman Sheikh, Christopher M Schafer, Christopher M West. J Biol Chem. 2022 Aug 3;102305. doi: 10.1016/j.jbc.2022.102305.
