NUDIX hydrolases with inorganic polyphosphate exo- and endo-polyphosphatase activities in the glycosome, cytosol and nucleus of Trypanosoma brucei
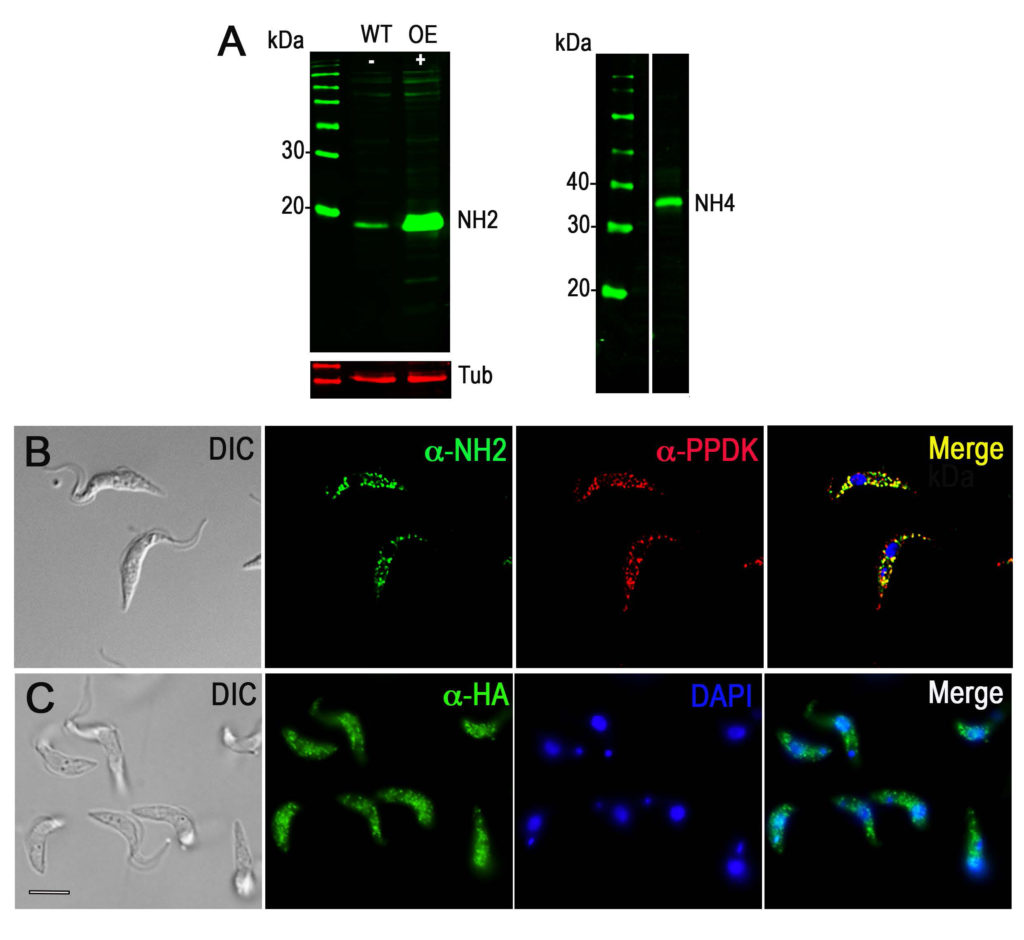 Trypanosoma brucei, a protist parasite that causes African trypanosomiasis or sleeping sickness, relies mainly on glycolysis for ATP production when in its mammalian host. Glycolysis occurs within a peroxisome-like organelle named the glycosome. Previous work from our laboratory reported the presence of significant amounts of inorganic polyphosphate (polyP), a polymer of three to hundreds of orthophosphate units, in the glycosomes and nucleoli of T. brucei In this work, we identified and characterized the activity of two Nudix hydrolases, TbNH2 and TbNH4, one located in the glycosomes and the other in the cytosol and nucleus, respectively, that can degrade polyP. We found that TbNH2 is an exopolyphosphatase with higher activity on short chain polyP, while TbNH4 is an endo- and exopolyphosphatase that has similar activity on polyP of various chain sizes. Both enzymes have higher activity at around pH 8.0. We also found that only TbNH2 can dephosphorylate ATP and ADP but with lower affinity than for polyP. Our results suggest that Nudix hydrolases can participate in polyP homeostasis and therefore may help control polyP levels in glycosomes, cytosol and nuclei of T. brucei.
Trypanosoma brucei, a protist parasite that causes African trypanosomiasis or sleeping sickness, relies mainly on glycolysis for ATP production when in its mammalian host. Glycolysis occurs within a peroxisome-like organelle named the glycosome. Previous work from our laboratory reported the presence of significant amounts of inorganic polyphosphate (polyP), a polymer of three to hundreds of orthophosphate units, in the glycosomes and nucleoli of T. brucei In this work, we identified and characterized the activity of two Nudix hydrolases, TbNH2 and TbNH4, one located in the glycosomes and the other in the cytosol and nucleus, respectively, that can degrade polyP. We found that TbNH2 is an exopolyphosphatase with higher activity on short chain polyP, while TbNH4 is an endo- and exopolyphosphatase that has similar activity on polyP of various chain sizes. Both enzymes have higher activity at around pH 8.0. We also found that only TbNH2 can dephosphorylate ATP and ADP but with lower affinity than for polyP. Our results suggest that Nudix hydrolases can participate in polyP homeostasis and therefore may help control polyP levels in glycosomes, cytosol and nuclei of T. brucei.
, , ,
