Researchers discover potential treatment for Chagas disease
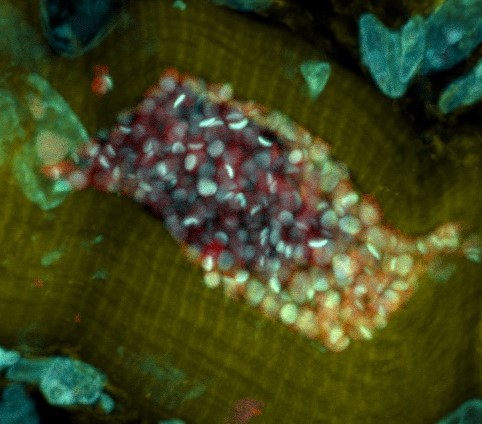
The skeletal muscle of a mouse infected with Trypanosoma cruzi is shown under a microscope. (Submitted by Fernando Sanchez) The condition affects tens of millions across the Americas but lacks effective treatments Researchers from the University of Georgia have discovered a potential treatment for Chagas disease, marking the first medication with promise to successfully and …