Cryopreservation of Plasmodium Sporozoites
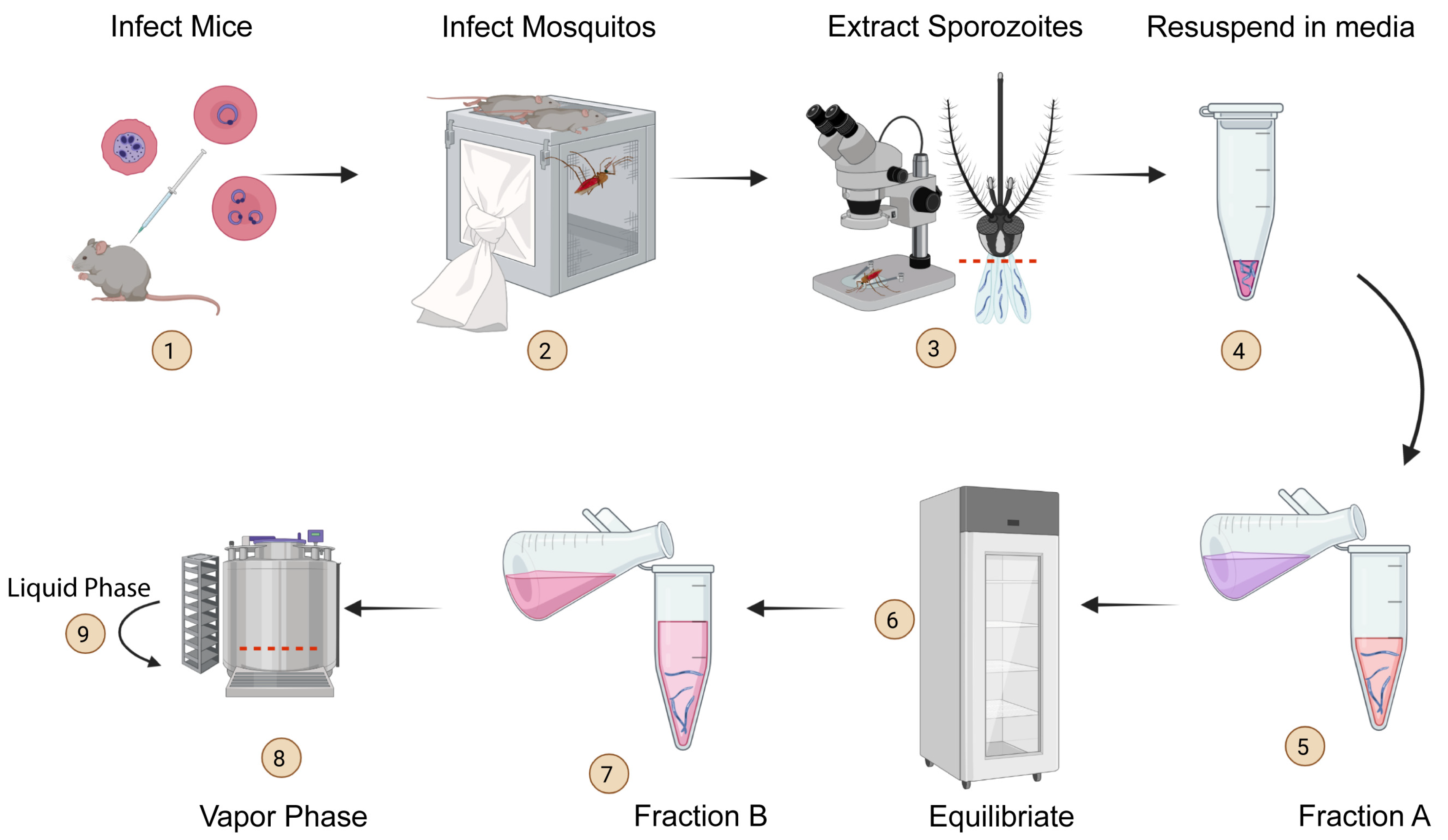
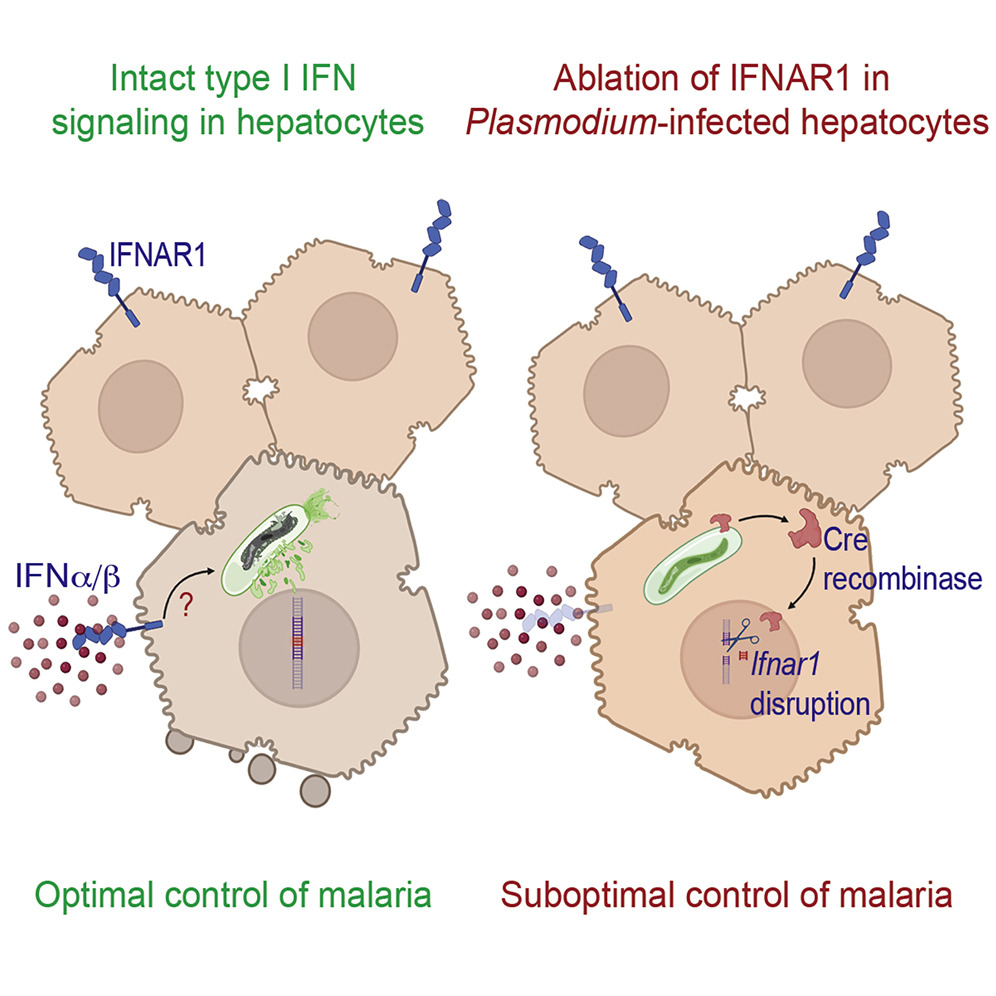
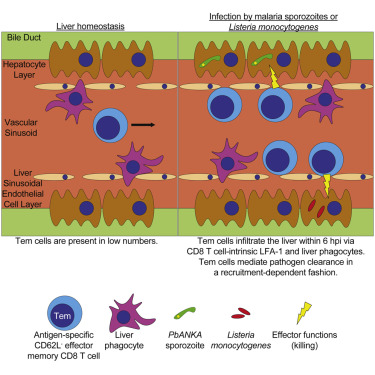
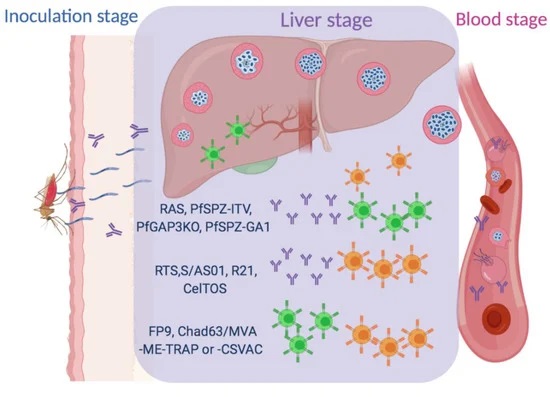
Cryopreservation protocol for Plasmodium sporozoites. Malaria is a deadly disease caused by the parasite, Plasmodium, and impacts the lives of millions of people around the world. Following inoculation into mammalian hosts by infected mosquitoes, the sporozoite stage of Plasmodium undergoes obligate development in the liver before infecting erythrocytes and causing clinical malaria. The most promising vaccine candidates for …