Mono-allelic epigenetic regulation of polycistronic transcription initiation by RNA polymerase II in Trypanosoma brucei

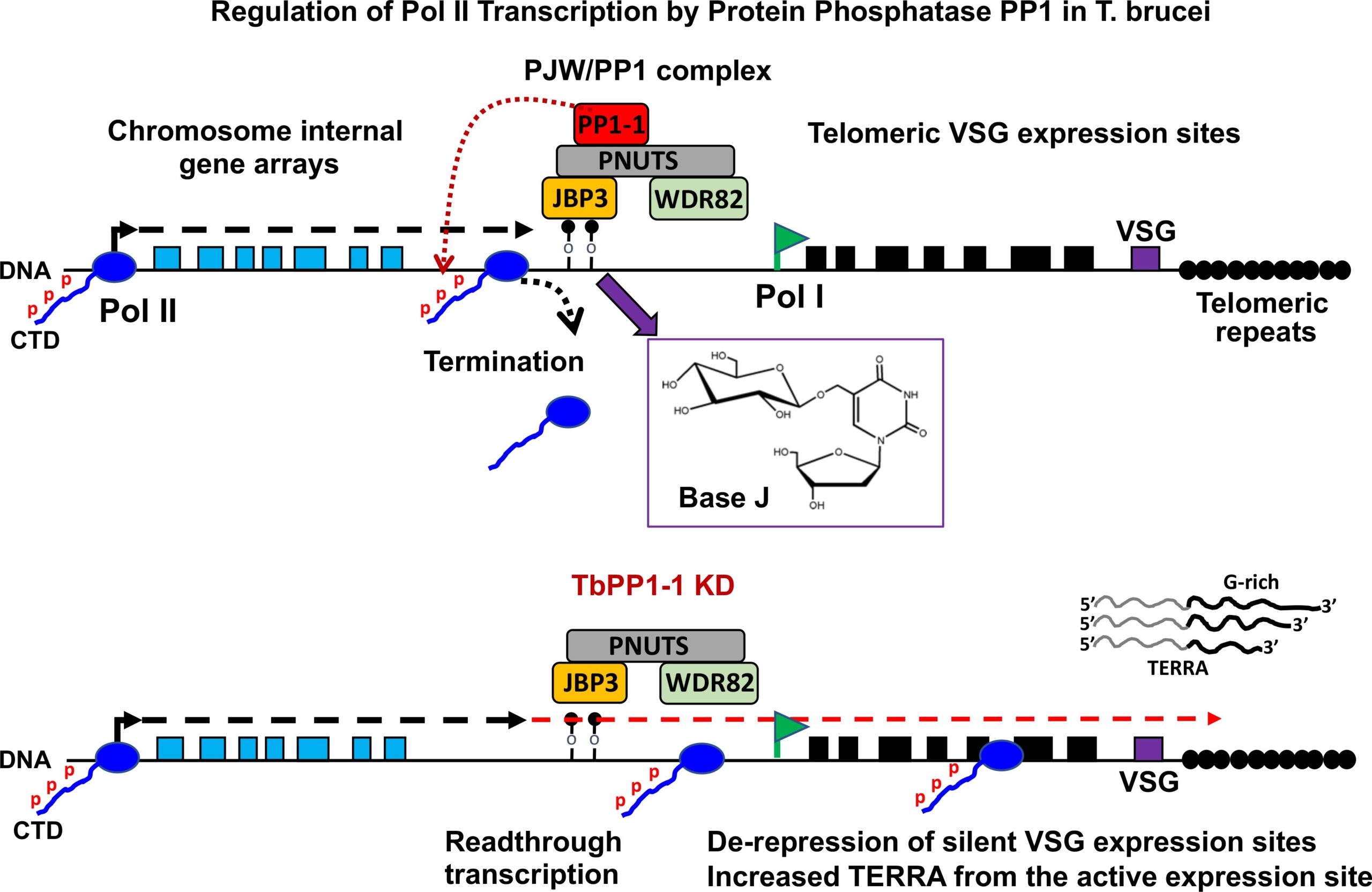
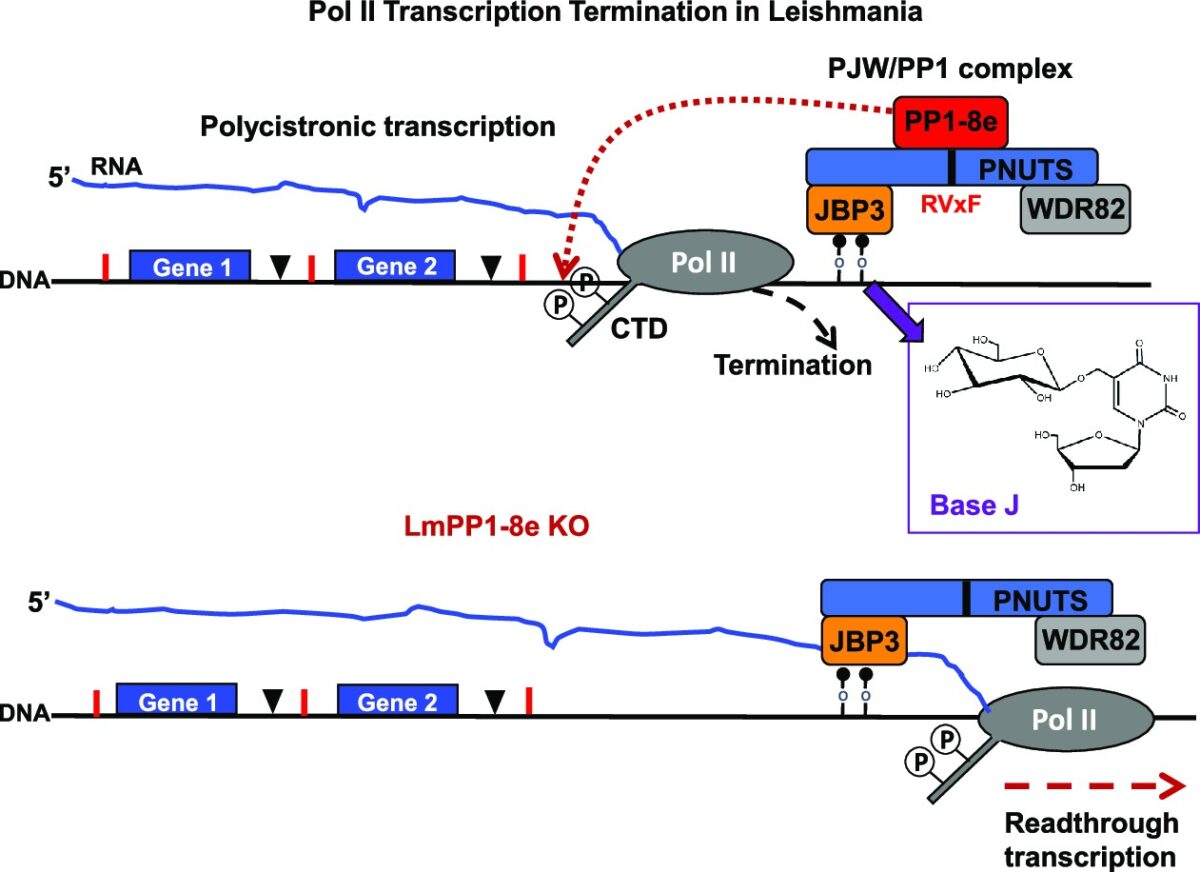
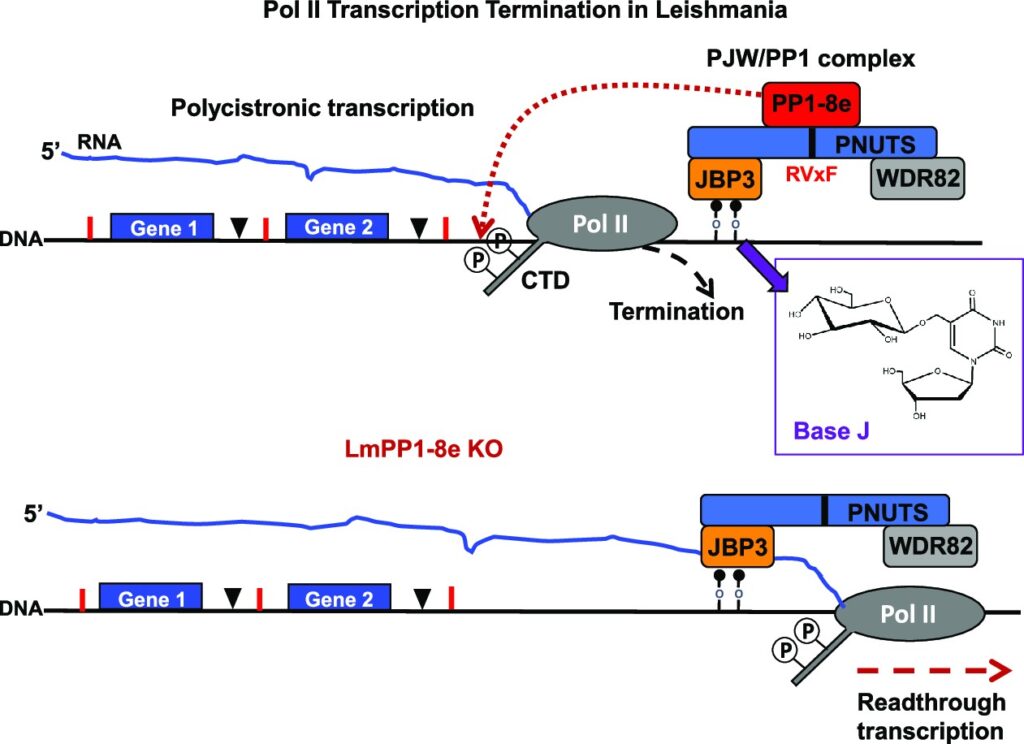
Unique for a eukaryote, protein-coding genes in trypanosomes are arranged in polycistronic transcription units (PTUs). This genome arrangement has led to a model where Pol II transcription of PTUs is unregulated and changes in gene expression are entirely post-transcriptional. Trypanosoma brucei brucei is unable to infect humans because of its susceptibility to an innate immune complex, trypanosome lytic factor (TLF) in the circulation of humans. The initial step in TLF-mediated lysis of T.b.brucei requires high affinity haptoglobin/hemoglobin receptor (HpHbR) binding. Here, we demonstrate that by in vitro selection with TLF, resistance is obtained in a stepwise process correlating with loss of HpHbR expression at an allelic level. RNA-seq, Pol II ChIP, and run-on analysis indicate HpHbR silencing is at the transcriptional level, where loss of Pol II binding at the promoter region specifically shuts down transcription of the HpHbR-containing gene cluster and the adjacent opposing gene cluster. Reversible transcriptional silencing of the divergent PTUs correlates with DNA base J modification of the shared promoter region. Base J function in establishing transcriptional silencing, rather than maintenance, is suggested by the maintenance of PTU silencing following the inhibition of J-biosynthesis and subsequent loss of the modified DNA base. Therefore, we show that epigenetic mechanisms exist to regulate gene expression via Pol II transcription initiation of gene clusters in a mono-allelic fashion. These findings suggest epigenetic chromatin-based regulation of gene expression is deeply conserved among eukaryotes, including early divergent eukaryotes that rely on polycistronic transcription.IMPORTANCEThe single-cell parasite Trypanosoma brucei causes lethal diseases in both humans and livestock. T. brucei undergoes multiple developmental changes to adapt in different environments during its digenetic life cycle. With protein-coding genes organized as polycistronic transcription and apparent absence of promoter-mediated regulation of transcription initiation, it is believed that developmental gene regulation in trypanosomes is essentially post-transcriptional. In this study, we found reversible Pol II transcriptional silencing of two adjacent polycistronic gene arrays that correlate with the novel DNA base J modification of the shared promoter region. Our findings support epigenetic regulation of Pol II transcription initiation as a viable mechanism of gene expression control in T. brucei. This has implications for our understanding how trypanosomes utilize polycistronic genome organization to regulate gene expression during its life cycle.
Rudo Kieft, Laura Cliffe, Haidong Yan, Robert J Schmitz, Stephen L Hajduk, Robert Sabatini. mBio. 2024 Dec 20:e0232824. doi: 10.1128/mbio.02328-24.