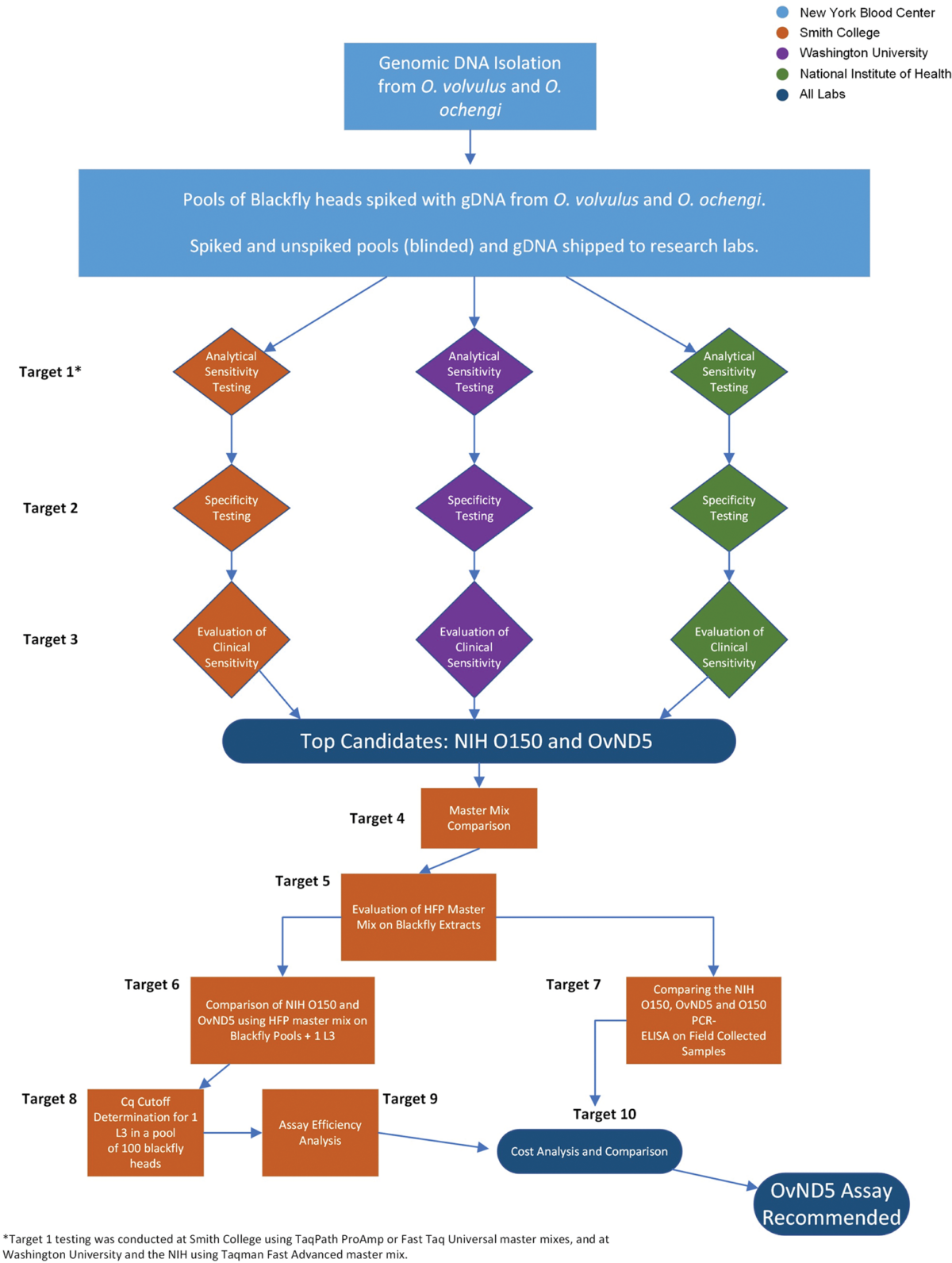
Optimized strategy for real-time qPCR detection of Onchocerca volvulus DNA in pooled Simulium sp. blackfly vectors
Background: Onchocerca volvulus is a filarial parasite that is a major cause of dermatitis and blindness in endemic regions primarily in sub-Saharan Africa. Widespread efforts to control the disease caused by O. volvulus infection (onchocerciasis) began in 1974 and in recent years, following successful elimination of transmission in much of the Americas, the focus …