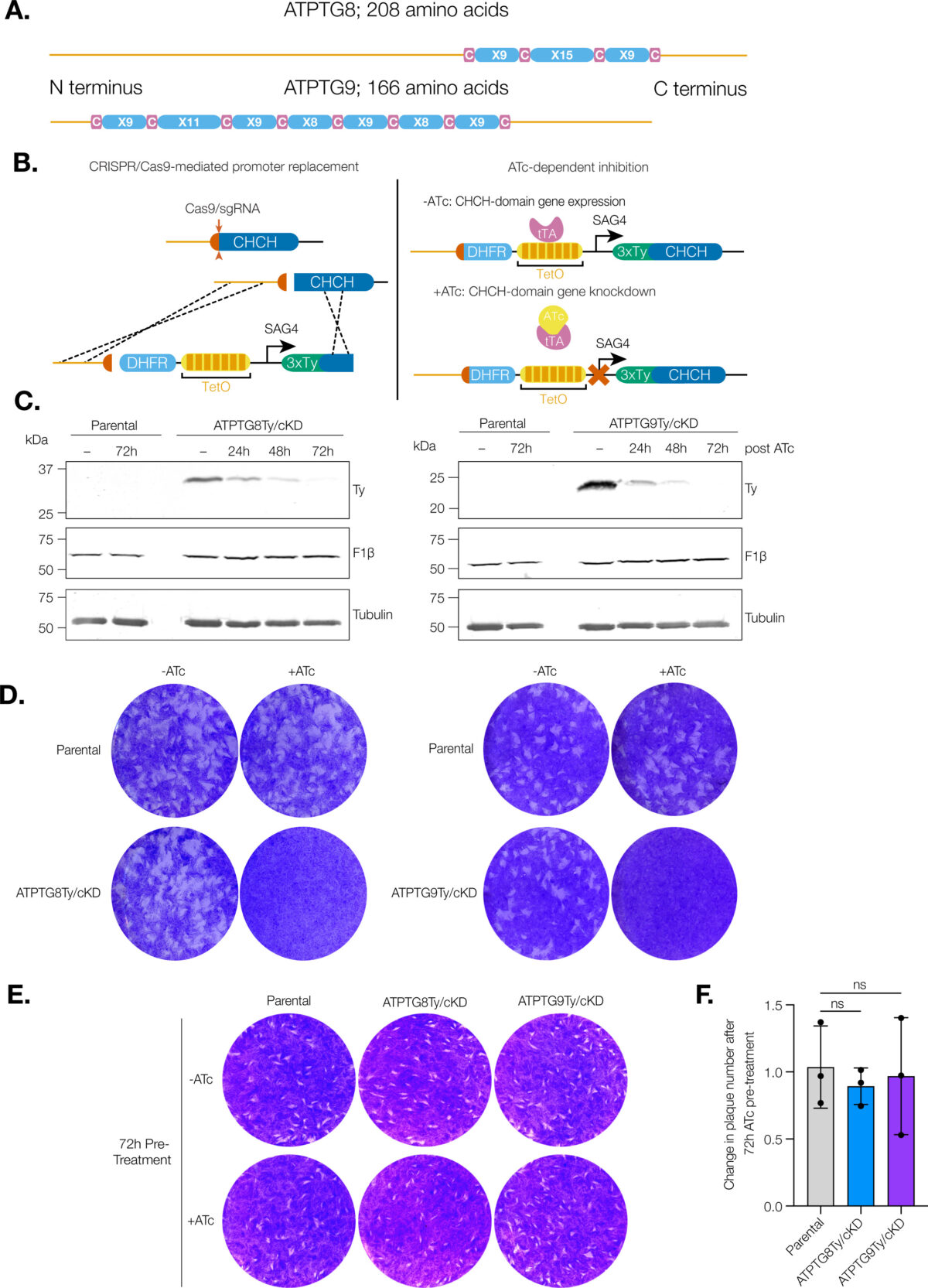
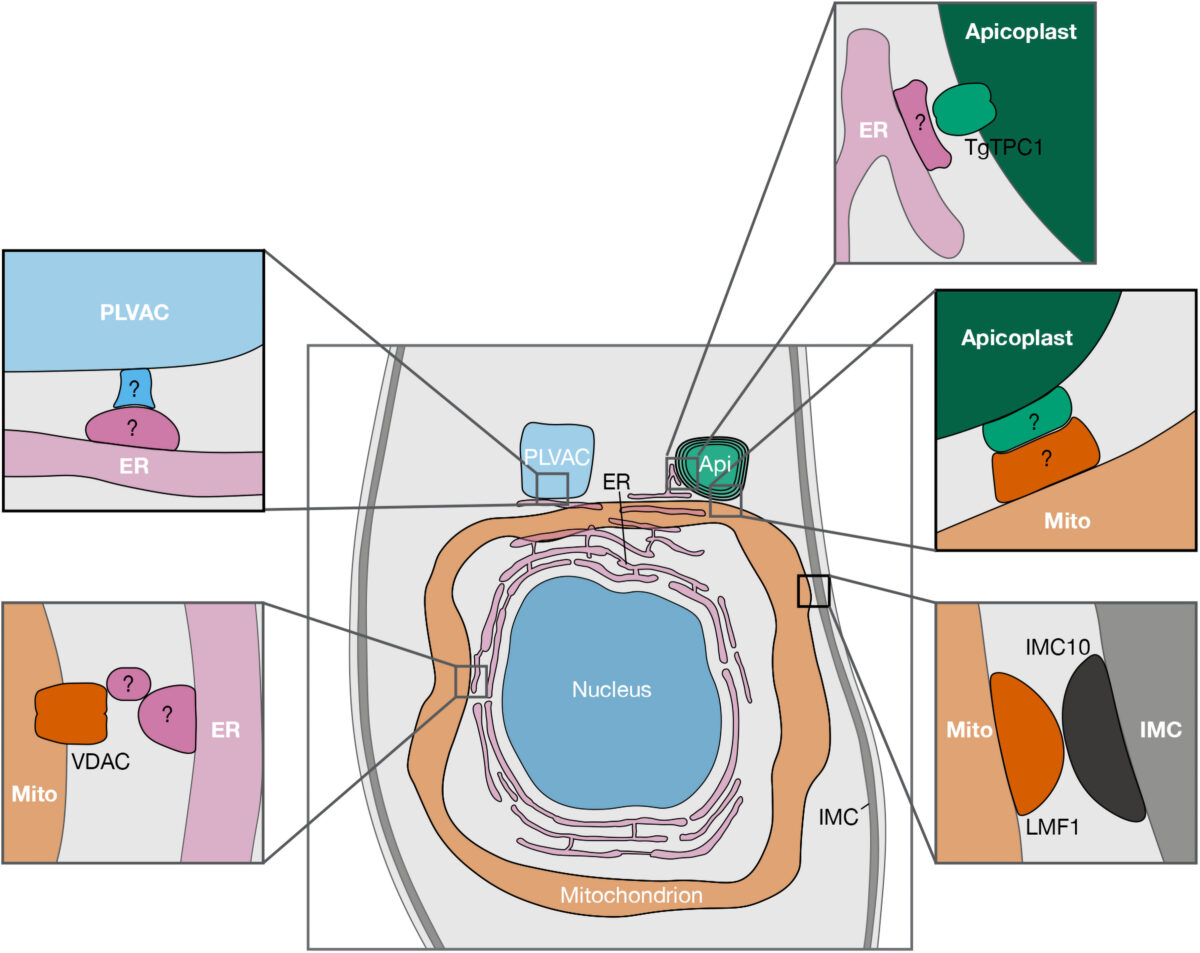
mSphere of Influence: Lighting up organellar communication in protozoan parasites
Diego Huet works in molecular parasitology, focusing on the organellar biology of Toxoplasma gondii. In this mSphere of Influence article, he reflects on how the article “Efficient proximity labeling in living cells and organisms with turboID” (Branon et al., 2018) impacted his research and the strategies used to dissect inter-organellar interactions in T. gondii. Diego …