Evaluation of changes in drug susceptibility and population genetic structure in Haemonchus contortus following worm replacement as a means to reverse the impact of multiple-anthelmintic resistance on a sheep farm
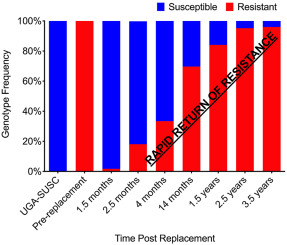
A population of Haemonchus contortus that was highly resistant to benzimidazoles and avermectin/milbemycins with a subpopulation that was resistant to levamisole, was replaced with a susceptible laboratory isolate of H. contortus in a flock of sheep. The anthelmintic susceptibility and population genetics of the newly established population were evaluated for 3.5 years using in vivo, in vitro, and molecular methods. Successful replacement of the resistant population with a susceptible population was confirmed using phenotypic and genotypic measurements; larval development assay indicated full anthelmintic susceptibility; albendazole treatment yielded 98.7% fecal egg count reduction; pyrosequence genotyping of single nucleotide polymorphisms in positions 167 and 200 of the isotype-1 beta tubulin gene were present at 0.0 and 1.7%, respectively; microsatellite genotyping indicated the background haplotype was similar to the susceptible isolate; and haplotypes of the isotype-1 beta tubulin gene were similar to the susceptible isolate. To sustain the susceptibility of the new population, targeted selective treatment was implemented using albendazole. Surprisingly, within 1.5 years post-replacement, the population reverted to a resistant phenotype. Resistance to albendazole, ivermectin, and moxidectin was confirmed via fecal egg count reduction test, larval development assay, and pyrosequencing-based genotyping. Targeted selective treatment was then carried out using levamisole. However, within one year, resistance was detected to levamisole. Population genetics demonstrated a gradual change in the genetic structure of the population until the final population was similar to the initial resistant population. Genetic analyses showed a lack of diversity in the susceptible isolate, suggesting the susceptible isolate had reduced environmental fitness compared to the resistant population, providing a possible explanation for the rapid reversion to resistance. This work demonstrates the power of combining molecular, in vitro, and in vivo assays to study phenotypic and genotypic changes in a field population of nematodes, enabling improved insights into the epidemiology of anthelmintic resistance.
Melissa M George, Adriano F Vatta, Sue B Howell, Bob E Storey, Ciaran J McCoy, Adrian J Wolstenholme, Elizabeth M Redman, John S Gilleard, Ray M Kaplan. International Journal for Parasitology: Drugs and Drug Resistance. 15, April 2021, 134-143. https://doi.org/10.1016/j.ijpddr.2021.02.004